Of course, we just as easily could have illustrated the law by considering the mass of nitrogen that combines with one gram of oxygen it works both ways!.Note that just as the law of multiple proportions says, the weight of oxygen that combines with unit weight of nitrogen work out to small integers there is a typo in Line 3-where there is a 3, there should be a 4. 
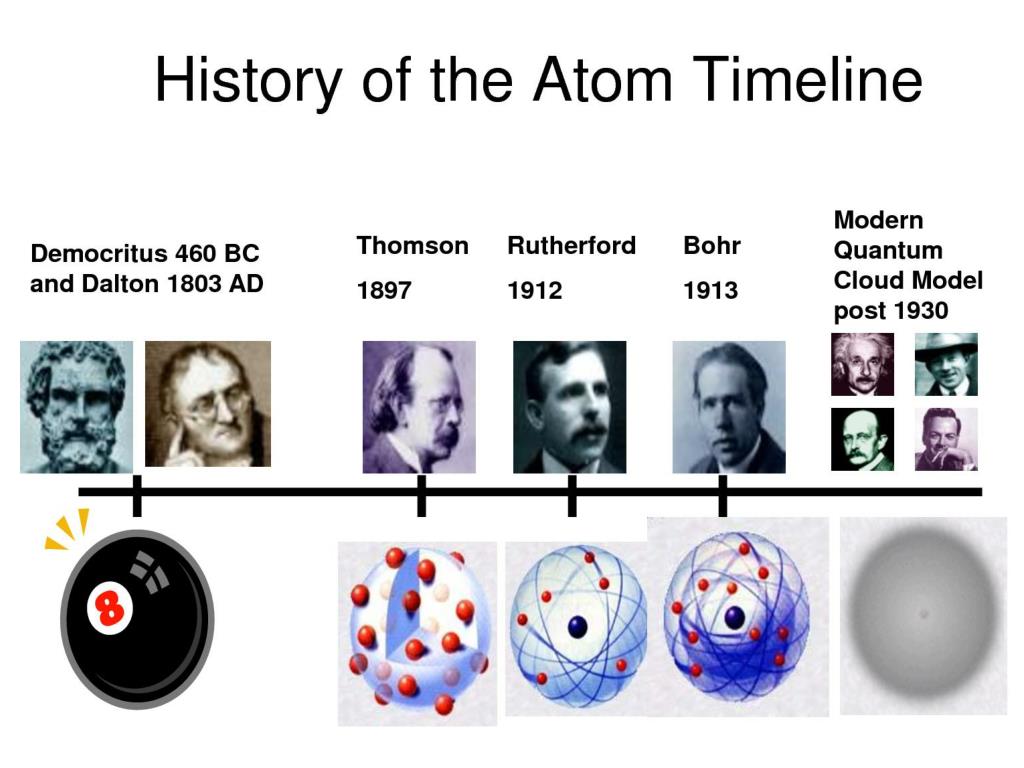
Line 3 is obtained by dividing the figures of the Line 2 by the smallest O:N ratio in Line 2 (which is the ratio for N 2O).But someone who depends solely on experiment would work these out by finding the mass of O that combines with unit mass (1 g) of nitrogen. The numbers in Line 2 are just the mass ratios of O:N, found by dividing the corresponding ratios in line 1.(These numbers were not known in the early days of Chemistry because atomic weights of most elements were not reliably known.) These ratios were calculated by simply taking the molar mass of each element, and multiplying by the number of atoms of that element per mole of the compound. Line 1 shows the ratio of the relative weights of the two elements in each compound.Overall, the development of views about atoms and the emergence of alchemy contributed to the evolution of modern chemistry, which now explores the fundamental building blocks of matter and the chemical reactions that occur between them.\)) compounds. Thomson discovered the electron, a subatomic particle that orbits the nucleus of an atom.ġ911: Ernest Rutherford conducted the gold foil experiment, which showed that atoms were mostly empty space with a dense, positively charged nucleus.ġ932: James Chadwick discovered the neutron, a subatomic particle that has no charge but contributes to the mass of an atom.ġ961: Linus Pauling proposed the concept of hybridization, which explained how orbitals in an atom can combine to form new hybrid orbitals that can accommodate bonding with other atoms.Ģ012: The discovery of the Higgs boson particle confirmed the existence of the Higgs field, which is responsible for giving particles mass and is considered a fundamental part of the standard model of particle physics. Timeline of the Development of Views about Atoms and Alchemy into Modern Chemistry:Ĥ00 BCE: Ancient Greek philosopher Democritus introduced the idea of atoms as indivisible particles that make up all matter.ģ00 BCE: Aristotle rejected Democritus' idea of atoms and proposed that all matter was made up of four elements: earth, air, fire, and water.Ĩ00 CE: Alchemy emerged as a philosophical and experimental practice that sought to transform base metals into gold and discover the philosopher's stone, a substance that could grant immortality.ġ661: Robert Boyle conducted experiments on gases, which led him to propose the concept of the "corpuscular philosophy" of matter, which stated that matter was made up of particles that obeyed natural laws.ġ803: John Dalton published his atomic theory, which stated that all matter was made up of tiny, indivisible particles called atoms.ġ869: Dmitri Mendeleev created the periodic table of elements, which organized elements by their atomic weight and allowed for the prediction of the properties of undiscovered elements.ġ897: J.J.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
